News All News
July 11th, 2018
Kidney podocytes, all grown up
New protocol produces large numbers of mature human podocytes, enabling kidney disease modeling, drug discovery, and potential cell therapies
By Lindsay Brownell, Wyss Institute
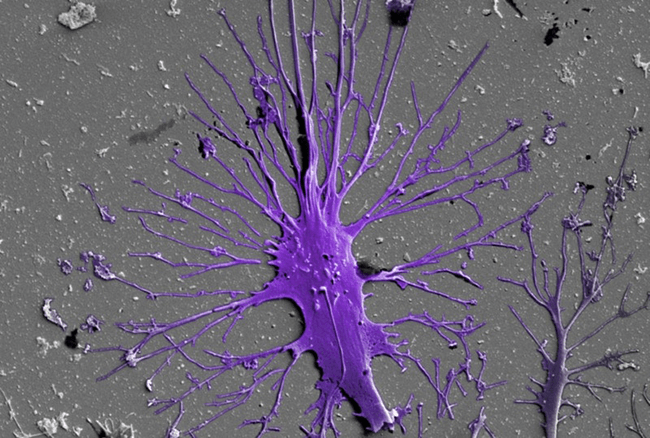
A mature podocyte, colored purple, grows long extensions of its cell body known as “feet” that wrap around the kidney’s blood vessels and help filter the body’s waste from the blood. (Image credit: Wyss Institute at Harvard University.)
July 11, 2018 - Human stem cells are of great interest in the fields of regenerative medicine and medical research because they reproduce indefinitely and can differentiate into every other cell type found in the body. While stem cells naturally occur in very few places in the adult body, induced pluripotent stem cells (iPS cells) can be produced directly from adult cells, and offer the potential for a patient to one day have a limitless source of personalized cells to replace those lost to damage or disease.
Previous work from the Wyss Institute at Harvard University established a protocol for producing human kidney podocytes (a type of cell that helps filter blood in the kidneys) from iPS cells with greater than 90% efficiency, and used those podocytes in a Glomerulus Chip that can recreate in vitro the specialized tissue structure and molecular filtration found in the glomerulus of the human kidney.
Now, using that protocol, Wyss researchers have shown that the differentiated cells exhibit transcriptomic and protein expression profiles that match those of mature podocytes – a feat that no other method has so far been able to achieve. This confirmation of mature podocytes gives kidney researchers across the scientific community a tool for investigating human kidney development, function, and disease; these cells also could potentially be delivered as a cell therapy for kidney diseases in the future. The research is reported in Nature Protocols.

Samira Musah, Ph.D., a Dean's Postdoctoral Fellow at Harvard Medical School and first author of the study, holds a Kidney Chip containing endothelial and glomerular cells. (Image credit: Wyss Institute at Harvard University.)
When iPS cells set out to differentiate, they first turn into “progenitor” cells, which only have the ability to form a subset of cell types. Progenitors that are destined to become podocytes first differentiate into nephron progenitor-like cells. While these cells can be used as a proxy for human cells in research, mature cells are of much greater use to researchers and clinicians, as they more closely mimic the cells found in adult organs. In order to produce mature podocytes, Samira Musah, Ph.D., a former Postdoctoral Fellow in the laboratory of Founding Director Donald Ingber, M.D., Ph.D., who is currently an Assistant Professor of Biomedical Engineering at Duke University, first created nephron progenitor-like cells from iPS cells, and then exposed them to a new cell culture medium containing a cocktail of five molecules that had been previously shown to play key roles in kidney development and function in vivo. This in vitro differentiation protocol resulted in cells that grew the long “foot” processes and expressed the genes that are the hallmarks of mature podocytes.
“Our method’s ability to produce mature human podocytes from iPS cells with high yield and without the need for subpopulation selection or genetic manipulations offers researchers and clinicians a robust, renewable source of kidney cells for scientific and medical studies,” said Musah, who is also a Dean’s Postdoctoral Fellow at Harvard Medical School.
There are many valuable uses for this protocol of mature podocyte generation, including investigating the steps involved in the differentiation of podocytes from their progenitor cells (a process that remains poorly understood), studying the origin and progression of various kidney diseases including podocytopathies and glomerulosclerosis, and establishing in vitro systems for kidney drug testing and discovery, such as the Glomerulus Chip. The researchers also hypothesize that mature human iPS cell-derived podocytes could one day be used as an injectable form of cell therapy for diseases that are characterized by podocyte loss or dysfunction.
“This method for generating mature human kidney podocytes should be of great value to basic researchers and investigators in regenerative medicine who are interested in studying various types of kidney disease or trying to develop cell-based therapies,” said Ingber, who is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and the Vascular Biology Program at Boston Children’s Hospital, as well as Professor of Bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.
Additional authors of the paper include Nikolaos Dimitrakakis, M.Eng, M.Sc, an Associate Biostatistician and Data Analyst at the Wyss Institute; Diogo Camacho, Ph.D., a Senior Bioinformatics Scientist at the Wyss Institute; and George Church, Ph.D., Core Faculty member of the Wyss Institute who is also a Professor of Genetics at Harvard Medical School and Professor of Health Sciences and Technology at Harvard and MIT.
This work was supported by DARPA, The Wyss Institute at Harvard University, Harvard Medical School, UNCF-Merck, the Burroughs Wellcome Fund, and the NIH-NIDDK.
Tags: bioengineering , stem cells
Press Contact: Kirsten Mabry | (617) 495-4157
